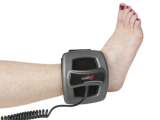
Description
CMF OL1000 Bone Growth Stimulators are portable, battery-powered medical devices indicated for use in the noninvasive treatment of an established nonunion fracture acquired secondary to trauma, excluding all vertebrae and flat bones. The device has the following features:
- Lightweight and comfortable
- Easy-to-use & Noninvasive
- Requires simple, one-button operation
- Device is worn for only 30 minutes per day
- Can be used with internal or external fixation or over a cast
This treatment has been shown in pre-clinical studies to help the body’s own healing process begin working. Clinical studies have shown an increased chance of healing of 60.7% in patients with nonunions that averaged 29.3 months from injury. Registry data on over 2300 patients reports a heal rate of 75.1%.
BRIEF PRESCRIBING INFORMATION
CONTRAINDICATIONS: Use of this device is contraindicated in individuals having a synovial pseudarthrosis. Demand-type pacemaker or implantable cardioverter defibrillator (ICD) operation may be adversely affected by exposure to magnetic fields. Physicians should not prescribe CMF 0L1000 for applications that may place the treatment transducers in close proximity to the pacemaker. Further screening by the attending cardiologist is recommended (such as with an electrocardiogram). CMF 0L1000 should not be used in the presence of external or internal fixation devices that are constructed from magnetic materials.
(NOTE: Almost all fracture fixation devices implanted today are made from nonmagnetic materials.)
WARNINGS: The safety and effectiveness of the use of this device on individuals lacking skeletal maturity have not been established. Animal studies conducted to date do not suggest any long-term significant adverse effects from use of this device. However, long-term effects in humans are unknown. Teratological studies have not been performed with this device. The safety of use of this device during pregnancy or nursing in humans has not been established.
PRECAUTIONS: Weight bearing is not advised in the presence of extreme motion at the nonunion site. In the presence of a malaligned nonunion, careful consideration of the use of this device must be undertaken on an individual basis, as treatment with this device is not intended to alter or affect the degree of malalignment. The safety and effectiveness of the use of this device on individuals with nonunion secondary to, or in conjunction with, a pathological condition have not been established. This device should not be used if there are mental or physical conditions that preclude patient compliance with the physician and device instructions. When conditions of atrophy are present or when fractures have remained unhealed for long periods of time, there may be less successful results.
ADVERSE EFFECTS: No known significant adverse effects have resulted from the use of this device. Clinical studies, animal studies, and tissue culture experiments conducted with the OL1000, which has the same treatment signal as the OL1000 SC1, have not indicated any evidence of significant adverse effects.
CAUTION: Federal law (U.S.A. and Canada) restricts this device to sale, distribution or use by or on the order of a physician.